how to draw lewis structures for compounds
4.three: Drawing Lewis Structures
- Page ID
- 165659
Learning Objectives
- To describe Lewis structures.
- To recognize molecules that are likely to have multiple covalent bonds.
DRAWING LEWIS STRUCTURES
For very elementary molecules and molecular ions, nosotros can write the Lewis structures by merely pairing up the unpaired electrons on the constituent atoms. See these examples:
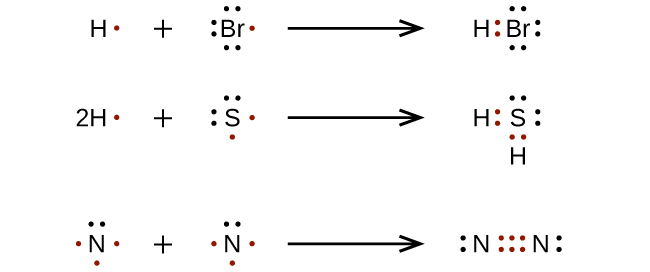
For more complicated molecules and molecular ions, it is helpful to follow the footstep-by-step procedure outlined here:
- Determine the total number of valence (outer beat) electrons among all the atoms. For cations, subtract one electron for each positive charge. For anions, add one electron for each negative accuse.
- Describe a skeleton construction of the molecule or ion, arranging the atoms around a central atom. (Mostly, the least electronegative element should be placed in the center.) Connect each atom to the central atom with a single bond (one electron pair).
- Distribute the remaining electrons as alone pairs on the terminal atoms (except hydrogen), completing an octet around each atom.
- Place all remaining electrons on the key atom.
- Rearrange the electrons of the outer atoms to brand multiple bonds with the central atom in order to obtain octets wherever possible.
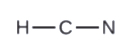
Allow us determine the Lewis structures of OF2 and HCN every bit examples in following this procedure:
1. Determine the total number of valence (outer shell) electrons in the molecule or ion. For a molecule, we add the number of valence electrons (utilise the master group number) on each atom in the molecule. This is the full number of electrons that must be used in the Lewis structure.
O + 2 (F) = OF2
6e- + (2 ten 7e-) = 20e-
H + C + Due north = HCN
1e-+ 4e-+ 5e-= 10e-
2. Draw a skeleton structure of the molecule or ion, arranging the atoms around a central atom and connecting each atom to the key cantlet with a single (i electron pair) bond. Notation that H and F can just form one bond, and are ever on the periphery rather than the primal atom.
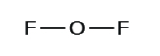
3. Distribute the remaining electrons every bit lone pairs on the final atoms (except hydrogen) to complete their valence shells with an octet of electrons.
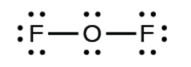
- In OFii, 6 electrons are placed on each F.
- In HCN, six electrons placed on N
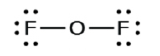
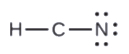
4. Place all remaining electrons on the central atom.
- In OFtwo, 4 electrons are placed on O.
- In HCN: no electrons remain (the total valence of 10e-is reached) and so naught changes.
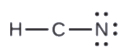
5. Rearrange the electrons of the outer atoms to brand multiple bonds with the central cantlet in society to obtain octets wherever possible.
- In OF2, each atom has an octet equally drawn, so nothing changes.
- In HCN, form ii more C–N bonds

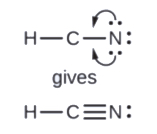
Finally, check to see if the full number of valence electrons are present in the Lewis construction. And then, inspect if the H atom has 2 electrons surrounding it and if each of the main group atoms is surrounded by 8 electrons.
MULTIPLE BONDS
In many molecules, the octet rule would not be satisfied if each pair of bonded atoms shares only ii electrons. Review HCN in Footstep 5 above. Some other example is carbon dioxide (CO2). CO2 has a total valence of 4e- + (ii x 6e-) = 16e-. Following steps 1 to iv, we describe the following:
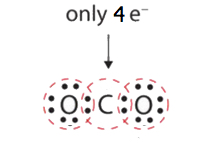
This does non give the carbon atom a complete octet; merely four electrons are in its valence shell. This organization of shared electrons is far from satisfactory.
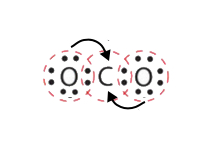
In this case, more than 1 pair of electrons must be shared betwixt two atoms for both atoms to have an octet. A second electron pair from each oxygen cantlet must be shared with the central carbon cantlet shown past the arrows in a higher place. A lone pair from each O must exist converted into a bonding pair of electrons.
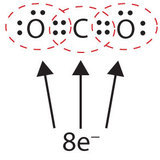
In this arrangement, the carbon atom shares four electrons (ii pairs) with the oxygen cantlet on the left and four electrons with the oxygen cantlet on the right. There are now eight electrons around each atom. Two pairs of electrons shared between 2 atoms make a double bond between the atoms, which is represented by a double dash:
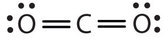
Some molecules contain triple bonds (like HCN, shown above). Triple bonds are covalent bonds in which three pairs of electrons are shared by two atoms. Another compound that has a triple bond is acetylene (C2H2), whose Lewis diagram is as follows:
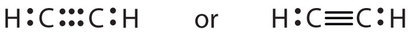
Case \(\PageIndex{1}\)
Describe the Lewis diagram for each molecule.
- \(\ce{N2}\)
- \(\ce{CH2O}\) (The carbon atom is the central atom.) One application of CHiiO, too called formaldehyde, is the preservation of biological specimens. Aqueous solutions of CHtwoO are chosen formalin and have a sharp, characteristic (pungent) smell.
Solution
- The total number of electrons is 2 ten 5 = x electrons. The bond between the ii nitrogen atoms is a triple bond. The Lewis diagram for North2is every bit follows:
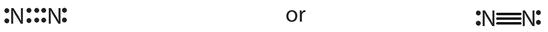
- The full number of electrons is four x 2(1) + 6 = 12 electrons. In CH2O, the central cantlet is surrounded by two different types of atoms. The Lewis diagram that fills each atom'due south valence electron shell is as follows:

Practice \(\PageIndex{1}\)
Depict the Lewis diagram for each molecule.
- \(\ce{O2}\)
- \(\ce{C2H4}\)
- Respond a:
-
 or
or 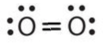
- Answer b:
-
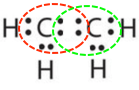 or
or 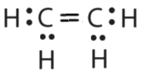 or
or 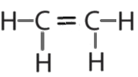
Example \(\PageIndex{2}\)
Which is the correct Lewis construction for NtwoH2?
A. 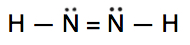
B. 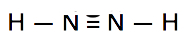
C. 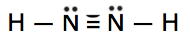
Solution
Lewis structure A is the correct reply. It has a total of (2 10 5e-) + (ii ten 1e-) = 12e-. Each of the N atoms satisfy the octet requirement and the H atoms follow the duet rule.
Construction B is electron deficient. Information technology has only 10e- instead of 12.
Structure C has xiv (2 extra) electrons. The N atoms practice not satisfy the octet.
Do \(\PageIndex{2}\)
Which is the correct Lewis structure for NOCl?
A. 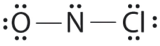
B. 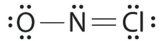
C. 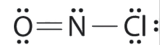
- Respond
-
Structure A violates the octet rule; Northward is surrounded past only 6e-.
Structure B violates the octet rule; Cl has 10e- around it. Furthermore, there are a total of 20e- instead of 18e-.
Structure C is the correct structure. Information technology has a total of 6e- + 5e- + 7e- = 18e-. Each atom is surrounded by 8 electrons (octet rule).
Primal Takeaways
- A Lewis construction shows the bonding and nonbonding electrons around individual atoms in a molecule.
- Some molecules must have multiple covalent bonds betwixt atoms to satisfy the octet rule.
- A double bail contains iv electrons and a triple bond contains vi electrons.
Exercises
-
What is one clue that a molecule has a multiple bond?
2. Describe the Lewis diagram for each of the following.
a. HtwoO
b. NH3
c. C2H6
d. CCl4
3. Each molecule contains double bonds. Draw the Lewis diagram for each. The first element is the central atom.
- CStwo
- CtwoFiv
- COCl2
iv. Each molecule contains multiple bonds. Draw the Lewis diagram for each. Assume that the first chemical element is the cardinal atom, unless otherwise noted.
- Northward2
- CO
- HCN (The carbon atom is the central atom.)
- POCl (The phosphorus cantlet is the primal atom.)
5. Explain why hydrogen atoms do not grade double bonds.
6. Why is it incorrect to draw a double bail in the Lewis diagram for MgO?
Answers
- If single bonds between all atoms practise not give all atoms (except hydrogen) an octet, multiple covalent bonds may be present.
- a.
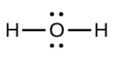
b.
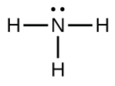
c.
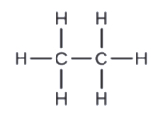
d.
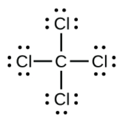
4. a. 
b. 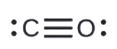
c. 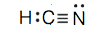
d.
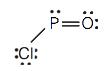
five. Hydrogen can accept only i more electron; multiple bonds require more than i electron pair to exist shared.
6. MgO is an ionic chemical compound (Mg transfers two electrons to O). The electrons are not shared hence it'southward incorrect to describe a double bond.
This is the Lewis dot structure of MgO.
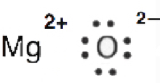
Source: https://chem.libretexts.org/Courses/Windward_Community_College/BIOC_141:_Fundamentals_of_Biochemistry_%28Colmenares_and_Ashburn%29/04:_Covalent_Bonding_and_Simple_Molecular_Compounds/4.3:_Drawing_Lewis_Structures
Posted by: pizzosament1964.blogspot.com


0 Response to "how to draw lewis structures for compounds"
Post a Comment